Lithium-ion batteries have received extensive attention from research workers and companies in various countries for their excellent overall performance. In the development process of lithium-ion batteries, electrode materials have become the bottleneck restricting the large-scale promotion and application of lithium-ion batteries, and the promotion and popularization of various small and medium-sized portable electronic products and electric bicycles (mobile phones, digital cameras, laptops, etc.) The commercial development of new-generation electric vehicles and hybrid vehicles puts higher requirements on the energy density and performance of lithium ion batteries, especially the traditional lithium cobalt oxide cathode materials and carbon anode materials in terms of specific capacity and specific energy. The new generation of lithium ion battery anode materials cannot meet the needs of the next generation of lithium ion battery anode materials. Therefore, the development of new high specific capacity, high stability, high safety, long life, low cost lithium ion battery electrodes is particularly urgent.
Lithium-ion battery electrodes are generally composed of a conductive agent, a binder, and an active material. Since the first-generation lithium-ion battery of Sony Corporation was introduced in the 1990s, the binder basically used polyvinylidene fluoride based on hydrogen bonding and van der Waals forces. Ethylene. With the use of large-capacity electrode materials (such as tin, silicon, etc.), this weak intermolecular force has not been sufficient to maintain the stability of the entire electrode system during charge and discharge.
Since the large-capacity electrode material faces extremely severe volume expansion (>200%) during charging, the strength of PVDF during volume expansion is insufficient to maintain the stability of the entire electrode system. Finally, the entire electrode system will There will be problems with the collapse of the bonding network, which will lead to a sharp drop in capacity and a decrease in the service life. Therefore, in recent years, improving the service life of large-capacity lithium-ion battery active materials has been a research hotspot in the scientific research industry and industry.
Institute of Nanotechnology and Nanomimicry, Suzhou Institute of Nanotechnology and Nanobionics, Chinese Academy of Sciences The Jianjian Research Group of Researchers differs from the traditional method of controlling the morphology of active materials. From the perspective of interface chemistry, the entire electrode system is focused on the entire electrode. The system has been designed to meet the needs of large capacity and long life. Firstly, a layer of nanometer-thickness (~10 nm) flexible polydopamine is coated on the surface of the active material as a buffer layer to release the deformation pressure caused by lithium ion intercalation; secondly, a negative pressure (<100 pa) solid-phase reaction is used in buffering. A covalent bond is formed between the layer and the binder to further increase the mechanical strength of the entire electrode system. For the first time in this study, a covalent bond was introduced into the electrode system. The mechanical strength of the electrode was approximately 70% lower than that of conventional electrodes based on hydrogen bonding and van der Waals force.
First of all, the research team conducted in-depth research and exploration of the most suitable buffer layer. Taking a tin-based negative electrode as an example, a protein-wrapped tin dioxide nanoparticle is prepared. Subsequently, in-situ encapsulation of polydopamine is performed on the surface of the nanoparticle. After being mixed with a carbon black conductive agent and a polyacrylic binder, negative pressure is applied. The in-situ cross-linking reaction causes the polyacrylic acid adhesive to form an amide bond with the polydopamine buffer layer and enhances the mechanical strength of the electrode. The circulated electrodes were characterized to find out the optimum buffer layer amount and the key role that the covalent bond plays in the charge and discharge of the electrode (Fig. 1).
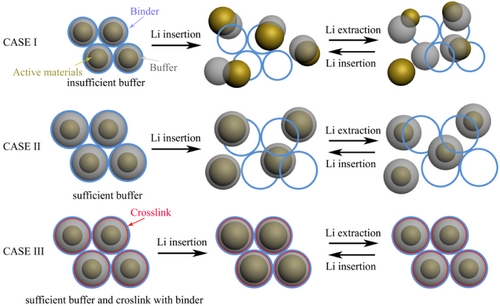
Figure 1. Schematic diagram of the role played by the buffer layer and cross-linking reaction during charge and discharge of Li-ion batteries.
Subsequently, the research team prepared a reduced graphene oxide/tin dioxide sheet structure to improve the conductivity of the electrode, and a layer of polydopamine layer was coated on the surface to cross-link with the binder (Figure 2). Intensity, this method greatly improves the service life and capacity of tin-based anode materials. After 200 cycles of charge and discharge, it is still possible to maintain a capacity of more than 700 milliampere hours, more than twice the theoretical value of the conventional carbon-based anode material.
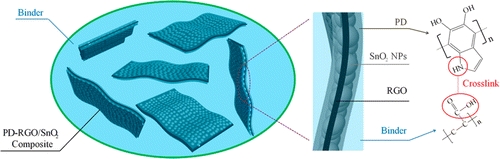
Figure 2. Polydopamine encapsulated reduced graphene and tin oxide complexes and their cross-linking mechanism
After improving the large-capacity negative electrode, the research team subsequently studied lithium-sulfur battery cathode materials. Lithium-sulfur battery is a new type of lithium-ion battery. Natural sulfur is used as a positive electrode material, and metallic lithium is used as a negative electrode. Because the cost of sulfur is extremely low, and its capacity is about 10 times that of conventional lithium cobalt oxide cathode material (1675 mAh/g), it is considered to be the most promising next-generation lithium-ion battery.
However, the conductivity of sulfur is extremely poor, and the lithium polysulfide formed during the charging process flows to the negative electrode, resulting in serious deterioration of the battery capacity. At the same time, as with other large-capacity active materials, sulfur also faces serious problems in the charging process. The problem of volume expansion. To solve the above problems, the research team first prepared reduced graphene oxide/sulfur composites to improve the overall conductivity of the material; secondly, the reduced graphene oxide/sulfur composites were coated with polydopamine to suppress the formation of the charging process. Lithium polysulfide overflow; Finally, the introduction of covalent bonds in the electrode system, increase the mechanical strength of the electrode, the life of the lithium-sulfur button battery is greatly improved than in the past, after the cycle 500 times, still remain near 700 mA/h capacity (Figure 3).
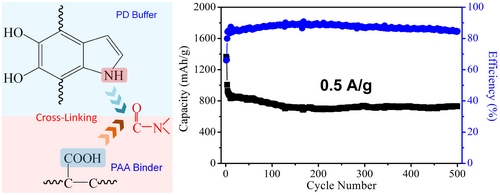
Figure 3. Cross-linking reaction of polydopamine buffer layer with polymer binder (left) and cycling performance (right).
Subsequently, on the basis of the above work, the research team used the milling method to prepare for the first time a sulphur nanosheet with a length of about 2 μm and a thickness of about 7 nm (Fig. 4), and replaced the conductive agent with a carboxylated multi-walled carbon nanotube. , In-situ cross-linking reactions were performed between the active material and the binder, the active material and the conductive agent, and a fully cross-linked lithium-sulfur battery cathode material was prepared for the first time (Fig. 5), wherein the strength of the interface between the conductive agent and the active material Nearly doubled. At a charge/discharge current of 1 amp/g, after 500 cycles, a high capacity was maintained, and the average capacity loss per cycle was only 0.021%, demonstrating an extremely stable electrode system.

Figure 4. Characterization of sulfur nanosheets by transmission electron microscopy (a, and b) and atomic force microscopy characterization (c).
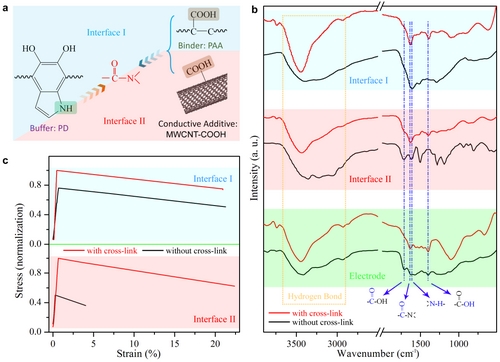
Figure 5. Schematic representation of fully cross-linked electrode interface characterization. a. Interface schematics; b. Infrared characterization of the cross-linked interface; c. Mechanical properties of the interface before and after cross-linking.
This series of work is different from the traditional lithium-ion battery research program. From the perspective of interface chemistry, the entire electrode system has been deeply studied and explored, and a series of designs have been made for each interface in the electrode. This method has a good general Adaptability, suitable for large-scale production, provides a new idea for the design and application of next-generation large-capacity lithium-ion batteries.
This series of work was supported by the national basic research projects (2013CB933000, 2010CB934700), the National Natural Science Foundation of China (21004076) and the major development project of the Chinese Academy of Sciences (KJZD-EW-M01-3). At the same time, Liu Zhaoping, a researcher at the Ningbo Institute of Materials, Chinese Academy of Sciences, and Chen Liji, a researcher at the Suzhou Nanotechnology Institute of the Chinese Academy of Sciences, both provided assistance in lithium ion battery testing and Li Qingwen's researchers in the pull test.
Pull Down Faucets,Pull Down Faucets Price,pull down kitchen faucet
Yuyao Zelin Sanitary Ware Co., Ltd , https://www.kitchen-sinkfaucet.com
